
WO 2015177807
Suryakant Shivaji Pol; Nitin Sharadchandra Pradhan; Shashikant Balu Padwal; Vihar Raghunath Telange; Nitn Shankar Bondre
Wanbury ltd

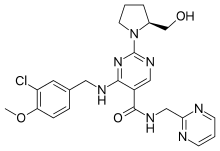
The present invention relates to a novel compound of Formula (II),
and its use in preparation of Avanafil, [Formula should be inserted
here] wherein R is -OH, -CI or -OR
1 and R
1 is C
1 to C
3 alkyl group
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2015177807&recNum=1&maxRec=&office=&prevFilter=&sortOption=&queryString=&tab=PCTDescription


It having been developed and launched by VIVUS and JW Pharmaceutical,
under license from Mitsubishi Tanabe Pharma, and Auxilium
Pharmaceuticals, for treating ED.
A process for preparation of Avanafil was first disclosed in US
6,797,709 (depicted in Scheme I), wherein
4-chloro-5-ethoxycarbonyl-2-methylthio-pyrimidine is coupled with
3-chloro-4-methoxybenzylamine in presence of triethylamine to provide
compound of Formula (A), which on oxidization provides a sulfonyl
compound of Formula (B). Said compound of Formula (B) is reacted with
L-prolinol and exert compound of Formula (C). The resulting compound of
Formula (C) undergoes column chromatographic purification and
crystallization, while further subjected to hydrolysis to obtain
compound of Formula (D). The compound of Formula (D) is coupled with
2-aminomethylpyrimidine to obtain Avanafil of Formula (I). The final
product obtained is purified by column chromatography. The need to
purify the intermediate compound of Formula (C) and final product, by
column chromatography makes this process cumbersome, time consuming and
unviable for large scale production thereby contributing to main
disadvantages of the process.
Scheme I

Formula (A)
m-CPBA/chloroform

Formula (C) Formula (B)
NaOH/DMSO

Formula (D) Formula (I)
CN 103254179, discloses a process for preparation of Avanafi, wherein
3-chloro-4-methoxybenzylhalide is coupled with cytosine to result
compound of Formula (E), later on condensation with L-prolinol yields
4-[(3-chloro-4-methoxy benzyl)amino-2-(2-hydroxymethyl)-l
-pyrrolinyl]pyrimidine of Formula (F). The compound of Formula (F) is
then condensed with N-(2-pyrimidylmethyl)formamide to obtain Avanafil of
Formula (I). Process is depicted in Scheme II
Scheme II

Formula (F) Formula (I)
CN 103254180 describes an alternate process for preparation of
Avanafil of Formula (I), wherein a substitution reaction on 6-amino-l
,2-dihydropyrimidine-2-keto-5-carboxylic acid, ethyl ester and
3-chloro-4-methoxybenzylchloride provides
6-(3-chloro-4-methoxybenzylamino)-l
,2-dihydropyrimidine-2-keto-5-carboxylic acid, ethyl ester of Formula
(G) which on condensation with L-prolinoI generates
6-(3-chloro-4-methoxybenzylamino)-l
,2-dihydropyrimidine-2-keto-5-carboxylic acid ethyl ester of Formula
(H). The compound of Formula (H) is then hydrolysed and coupled with
N-(2-pyrimidylmethyI)formamide to obtain Avanafil of Formula (I).
Process is depicted in Scheme III
Scheme III

Formula (H) Formula (Γ)
In all the prior art discussed above, chiral compound L-prolinol is
coupled in molecule in earlier steps of synthesis. This approach seems
to be less feasible for large scale production; the insertion of
L-prolinol in early stage may need to exert number of purifications for
intermediates. Further the main shortcoming in such process is that the
chirality of molecule is disturbed by inserting L-prolinol in early
stages because there are number of operations in line in process to
obtain the target compound.
CN 103483323, discloses a synthetic method for preparation of
avanafil, wherein amidation of pyrimidine-5-carbonyl chlorides with
2-(aminomethyl)pyrimidine at temperature ranging from -10 to 5°C
resulted an amide (intermediates A); which underwent condensation with
3-chloro-4-methoxybenzylamine at the temperature ranging from 0 -3°C to
give 4-[(3-chloro-4-methoxybenzyl)amino]-5-
pyrimidinecarboxamides (intermediates B), which further on
condensation with L-prolinol gave avanafil. The disadvantage of this
process is the need to maintain the reaction temperature in range of –
10 to 5°C which adds up to cost of process and makes the process
complicated. The process is depicted in Scheme IV.
Scheme IV

Intermediate (A)

wherein, R’ & R
2 are independently, hydrogen, halogen, alkoxy, alkoxyalkyl, cyno group, amino group
Hence, to overcome shortcomings of prior art the inventors of present
invention have skillfully designed a process with novel intermediate
which concomitantly result Avanafil compound of Formula (I),
substantially free from impurities. Further this invention encompass
L-proline in last stage of molecule in order to avoid the number of
purifications of intermediate which relent the economic significances by
taking into account yield of each stage.

Object of the invention
1. The main object of the invention is to provide a novel compound of Formula
(ID-
2. Another object of present invention is to provide a process for preparation of a novel compound of Formula (II).
3. Yet another object of present invention is to provide a process
for preparation of Avanafil of Formula (I), in high yield and purity
using a novel compound of Formula (II).
4. Yet another object of the present invention to provide simple,
economic and industrially scalable process for the preparation of
Avanafil o Formula (I).
Summary of the invention
According to an aspect of present invention, there is provided a novel compound of Formula (II).

Formula (II)
wherein R is -OH, -CI or -OR and R is Q to C
3 alkyl group
The invention will be specifically described below with reference to
Examples but it should not be construed that the scope of the invention
is limited thereto. Since the starting compound was produced by a
modified method from that described in prior art, it will be described
as Referential Example 1 to 3. Here synthesis routes of Referential
Example 1 to 3 and Example 1 to 10 are illustrated below in Scheme (V).
Scheme (V)

Formula (I) Referential Examples
Referential Example 1 – Preparation of ethyl 4-[(3-chloro-4-methoxybenzyl)amino]-2-(methyl sulfanyl)pyrimidine-5-carboxylate
To 600ml of methylene dichloride was added l OOg of ethyl
4-chloro-2-(methylsulfanyl) pyrimidine-5-carboxylate and 91.2g of
3-chloro-4-methoxybenzylamine. The reaction mixture was stirred and
500ml of water, 48g of sodium carbonate and Ig of tetra-butylammonium
bromide were added to it. The reaction mixture was then maintained
overnight at 25-30°C. After completion of reaction, methylene dichloride
layer was separated, washed with water and evaporated to obtain 145g of
ethyl 4-[(3-chloro-4-methoxybenzyl) amino]-2-(methyl sulfanyl)
pyrimidine-5-carboxylate having 95% of HPLC purity.
Above reaction can also be carried out using ammonia or triethylamine
in same reaction conditions and parameters, in place of sodium
carbonate.
Referential Example 2 – Preparation of 4-[(3-chloro-4-methoxybenzyl) amino]-2-(methyl sulfanyl) pyrimidine-5-carboxylic acid
To 600ml of methanol was added l OOg of ethyl
4-[(3-chloro-4-methoxybenzyl) amino]-2-(methyl sulfanyl)
pyrimidine-5-carboxylate (Referential Example 1) and an aqueous solution
of sodium hydroxide (15g of NaOH in 140ml of water). The reaction
mixture was heated to reflux temperature. After completion of reaction,
the pH of mixture was adjusted to 1 -2 using concentrated hydrochloric
acid followed by stirring the mixture for 1 hour at 10-15°C. The solid
product obtained was filtered, washed sequentially with water and
methanol, and dried overnight at 70-75°C to get 87g of
4-[(3-chloro-4-methoxybenzyl) amino]-2-(methyl sulfanyl)
pyrimidine-5-carboxylic acid.
Referential Example 3 – Preparation of 4-[(3-chloro-4-methoxybenzyl)
amino]-2-(methyl sulfinyl)-N-(pyrimidin-2-ylmethyl)
pyrimidine-5-carboxiamide of Formula (III)
To a mixture of 400ml of toluene and 0.5ml of dimethyl formamide was
added 50g of 4-[(3-chloro-4-methoxybenzyl) amino]-2-(methyl sulfanyl)
pyrimidine-5-carboxylic acid (Referential Example 2) and 70g of thionyl
chloride, and the reaction mixture was refluxed for 2.5 hours. After
completion of reaction, solvent was distilled under vacuum and the
residue was stripped with toluene to obtain yellow solid mass. The solid
mass thus obtained, was cooled to 15-20°C followed by addition of 1
75ml of methylene dichloride, 36. l g of 2-amino methyl pyrimidine
mesylate and 35.55g of triaethylamine. The reaction mixture was stirred
overnight at 25-30°C. After completion of reaction, methylene dichloride
was distilled out to get residue. The residue was washed sequentially
with 2.5% sodium carbonate solution and water. The residue was then
treated with methanol to obtain 4-[(3-chloro-4-methoxybenzyl)
amino]-2-(methyl sulfinyl)-N-(pyrimidin-2-ylmethyl)
pyrimidine-5-carboxiamide of Formula (III) having HPLC purity of more
than 95% (yield: 80%)
Referential Example 4 – Preparation of
4-[(3-Chloro-4-methoxybenzyl)amino]-2-[(2S)-2-(hydroxymethyl)-l
-pyrrolidinyl]-N-(2-pyrimidinylmethyl)-5-pyrimidinecarboxamide
(Avanafil)
Step i)
To 200ml of dichloromethane was added lOg of
4-[(3-chloro-4-methoxybenzyl) amino]-2-(methyl
sulfinyI)-N-(pyrimidin-2-ylmethyl) pyrimidine-5-carboxiamide and 6.5g of
m-chloro per benzoic acid and the mixture was stirred for 1 hour at
25-30°C. After completion of reaction, the reaction mixture was washed
with aqueous solution of sodium carbonate and water. The resulting
dichloromethane layer comprising compound of Formula (IV) was taken to
next step.
Step ii)
To the dichloromethane layer obtained in step i), was added 2.57g of
triethylamine followed by slow addition of 125ml solution of L-prolinol
in dichloromethane (2.46g of L-prolinol in 125ml of dichlromethane). The
reaction mixture was maintained overnight. After completion of
reaction, the reaction mixture was washed with water followed by
evaporation of dichloromethane to obtain an oily mass. The oily mass
thus obtained was treated with methanol to yield 8g of Avanafil.
Examples
Example 1 : Preparation of Compound of Formula (II) (wherein R is -OH)
Step i)
To 200ml of methylene dichloride was added lOg of
4-[(3-chloro-4-methoxybenzyl) amino]-2-(methyl
sulfinyl)-N-(pyrimidin-2-ylmethyl) pyrimidine-5-carboxiamide of Formula
(III) and 6.5g of m-chloro per benzoic acid and the mixture was stirred
for 1 hour at 25-30°C. After completion of reaction, the reaction
mixture was washed with aqueous solution of sodium carbonate and water.
The resulting methylene dichloride layer comprising compound of Formula
(IV) was taken to next step.
Step ii)
To the methylene dichloride layer comprising compound of Formula (IV)
obtained in step i), was added 5g of triethylamine followed by slow
addition of 125ml solution of L-proline in methylene dichloride (2.8g of
L-proline in 125ml of methylene dichloride). The reaction mixture was
maintained overnight. After completion of reaction, the reaction mixture
was washed with water and 5% sodium carbonate solution, followed by
evaporation of methylene dichloride to obtain an oily mass. The oily
mass obtained was stripped with 50ml acetone to yield 9g of compound of
Formula (II) having HPLC purity 98%.
Example 2: Preparation of Compound of Formula (II) (wherein R is -OC
2H
5)
To 100ml of ethanol was added 0.5ml of sulphuric acid and l Og of
compound of Formula (II) obtained in example 1 , and the reaction
mixture was maintained at reflux temperature till completion of
reaction. The reaction mixture was then cooled to 25-30°C and the pH of
reaction mixture was adjusted to 7-8 using sodium carbonate. Filter the
reaction mixture and collect filtrate containing product. The ethanol in
filtrate is completely distilled out to isolate 10.45g of esterified
compound of Formula (II).
Example 3 : Preparation of Compound of Formula (II) (wherein R is -CI)
To a mixture of 400ml of toluene and 0.5ml of dimethylformamide was
added 50g of compound of Formula (II) obtained in example 1 , and 70g of
thionyl chloride. The reaction mixture was refluxed for 2.5 hours.
After completion of reaction, solvent was distilled under vacuum and the
residue was stripped with toluene to obtain 50.5g of oily carboxylic
acid chloride compound of Formula (II).
Example 4: Preparation of Avanafil of Formula (I)
In an inert atmosphere, a solution of 30g of compound of Formula (II)
obtained in example 1 or 2, in 150 ml of tetrahydrofuran was dropwise
added to 180ml of suspension of 1.0M lithium aluminium hydride solution
in tetrahydrofuran, The reaction mixture was refluxed for 5 hours. After
completion of reaction, the mixture was cooled in ice-bath and
saturated aqueous solution of sodium sulfate was added to decompose
excess of lithium aluminium hydride. The mixture was then diluted with
200ml of methylene dichloride and thus formed organic layer was
separated. The organic layer was washed with water (3
χ 100
ml), dried over MgS04 and concentrated to collect crude Avanafil of
Formula (I) which was subjected to purification using methanol as
solvent to yield 22.8g of Avanafil of Formula (I) having HPLC purity of
99.20%.
Example 5 : Preparation of Avanafil of Formula (I)
To a mixture of 1.3g sodium borohydride, 1 ml methanesulfonic acid
and 50ml ethanol was added l Og of compound of Formula (II) obtained in
example 1 or 2, and the mixture was stirred at 25-30°C for 5 hours.
After completion of reaction, 100ml water was added and the mixture was
extracted with 1 00ml methylene dichloride (50ml X 2). The methylene
dichloride layer obtained was evaporated under reduced pressure to get
an oily mass. The oily mass was stripped with ethyl acetate at 45- 50°C.
To the oily residue formed was added 50ml of ethyl acetate and the
mixture was cooled to 0-5°C. The solid obtained was filtered, washed
with ethyl acetate and dried to yield crude Avanafil of Formula (I)
which was subjected to purification using methanol as solvent to yield
7g of Avanafil of Formula (I) having HPLC purity of 99%.
Example 6 to Example 8
The procedure is carried out as in example 5 except for instead of
methanesulfonic acid other reducing agents are used in combination with
sodium borohydride. The results are given in Table I
Table I

Example 9: Preparation of Avanafil of Formula (I)
To 100ml of ethanol was added 0.5ml of sulphuric acid and l Og of
compound of Formula (II) obtained in example 1 , and the reaction
mixture was maintained at reflux temperature till completion of
reaction. The reaction mixture was then cooled to 25-30°C and the pH of
reaction mixture was adjusted to 7-8 using sodium carbonate. Filter the
reaction mixture and collect filterate containing product. To the fi
Iterate was added 1.2g of sodium borohydride and 2.6g of lithium
bromide, and the mixture was stirred for 5 hours. After complete
conversion of ester to final product, l OOml water was added and the
mixture was extracted with 100ml methylene dichloride (50ml X 2). The
methylene dichloride layer obtained was evaporated under reduced
pressure to get an oily mass. The oily mass was stripped with 25ml ethyl
acetate at 45-50°C. To the oily residue formed was added 50ml of ethyl
acetate and the mixture was cooled to 0-5°C. The solid obtained was
filtered, washed with ethyl acetate and dried to yield crude Avanafil of
Formula (I) which was subjected to purification using methanol as
solvent to yield 7.5g of Avanafil of Formula (I) having HPLC purity of
99%.
Example 10: Preparation of Avanafil of Formula (I) from Compound of Formula (II) (wherein R is -CI)
To a mixture of 400ml of tetrahydrofuran and 50g of carboxylic acid
chloride compound of Formula (II) obtained in example 3, was added 12g
sodium borohydride at 0-5°C. After completion of reaction, water was
added to reaction mixture to decompose excess of sodium borohydride
present. The reaction mixture was then concentrated and a solution of
30g of potassium hydroxide in 200 ml of water was added. The mixture was
heated to 60-70°C and maintained for 15-18 hours. The mixture was then
cooled to 25-30°C and 500 ml of methylene dichloride was added. The
organic layer thus formed, was separated and evaporated to yield crude
Avanafil
of Formula (I) which was then subjected to purification using
methanol as solvent to obtain 40g of Avanafil of Formula (I) having HPLC
purity of 99.01%.


| Mr. K. Chandran |
|
|
|
Wholetime Director & Vice Chairman |

Tarapur plant


MR K. CHANDRAN (left), Director, Wanbury, and Mr Asok Shinkar,
Director-Corporate Finance, at a press conference held in Mumbai on
Monday.
//////////////
































